Covering COVID-19 is a daily Poynter briefing of story ideas about the coronavirus and other timely topics for journalists, written by senior faculty Al Tompkins. Sign up here to have it delivered to your inbox every weekday morning.
Covering COVID-19 is a daily Poynter briefing of story ideas about the coronavirus and other timely topics for journalists, written by senior faculty Al Tompkins. Sign up here to have it delivered to your inbox every weekday morning.
Late Tuesday, The Washington Post reported that this week, the Food and Drug Administration will issue guidance that it hopes will build public trust in whatever vaccine emerges to fight COVID-19. The Post reported:
The FDA is expected to ask manufacturers to follow participants in late-stage clinical trials for a median of at least two months, starting after they receive a second vaccine shot, according to two individuals familiar with the situation who spoke on the condition of anonymity.
Kaiser Health News created a stir this week when it reported that the medical world is becoming increasing worried that President Donald Trump, who has criticized the Food and Drug Administration, could use his authority to assert an emergency authorization of a COVID vaccine, if such a vaccine became available, but not get FDA approval. Kaiser said:
If the executive branch were to overrule the FDA’s scientific judgment, a vaccine of limited efficacy and, worse, unknown side effects could be rushed to market. The worries intensified over the weekend, after Alex Azar, the administration’s secretary of Health and Human Services, asserted his agency’s rule-making authority over the FDA. HHS spokesperson Caitlin Oakley said Azar’s decision had no bearing on the vaccine approval process.
Azar’s office says it has no intent to do an end-run around the FDA and Azar “will defer completely to the FDA.”
“The idea the Secretary would approve or authorize a vaccine over the FDA’s objections is preposterous and betrays ignorance of the transparent process that we’re following for the development of the Operation Warp Speed vaccines,” HHS Chief of Staff Brian Harrison wrote in an email to Kaiser.
Kaiser pointed out that:
Vaccines are typically approved by the FDA. Alternatively, Azar — who reports directly to Trump — can issue an emergency use authorization, even before any vaccines have been shown to be safe and effective in late-stage clinical trials.
“Yes, this scenario is certainly possible legally and politically,” said Dr. Jerry Avorn, a professor of medicine at Harvard Medical School, who outlined such an event in the New England Journal of Medicine. He said it “seems frighteningly more plausible each day.”
The president has been able to bend the FDA to favor two treatments that lacked overwhelming evidence that they were safe and effective: hydroxychloroquine and convalescent plasma.
As this sits today, HHS and FDA say there will be no executive action to fast-track a vaccine. That’s as it sits today. As an old boss of mine used to say, “Stay close, stuff changes.”
Is a wave of COVID in Europe heading to America?
One thing I have learned from my Poynter colleagues at the International Fact-Checking Network is that when they see COVID-19 cases ticking up in Asia, then Europe, in a month or so we can count on seeing something similar in America.
Tuesday, I led a discussion with journalists from around the globe, from Sierra Leone, Japan, Bosnia, Liberia, Ireland, France and beyond. What I heard from these journalists is a persistent rise in COVID-19 cases, whether it is a “second wave” or just more of the first wave that never flattened. It is prompting strict new rules for businesses and gatherings and those — as you might imagine — are not popular notions to enforce.
The European Centre for Disease Prevention and Control said on Monday that Spain had 300 cases of COVID-19 per 100,000, the highest in Europe, followed by the Czech Republic, France, Luxembourg, Malta, Belgium, Hungary, and Austria.
There is also concern about rising cases in the UK and France.
In Spain, parts of the capital Madrid saw new lockdown measures enforced on Monday. More than 850,000 residents face restrictions on travel and the size of gatherings. On Tuesday, Spain reportedly requested the army’s help to enforce some of the restrictions.
In the Czech Republic, in a televised debate on Sunday, former health minister Adam Vojtech who resigned on Monday as cases climbed above 50,000 said the government could declare a state of emergency if the infection rate continued to rise — a measure several European countries took at the height of the pandemic.On September 12, French authorities reported 10,000 COVID-19 cases in one day for the first time since the pandemic began. Since then, daily cases have soared to more than 13,000. Several “red zones,” cities considered high risk, have seen clampdowns.
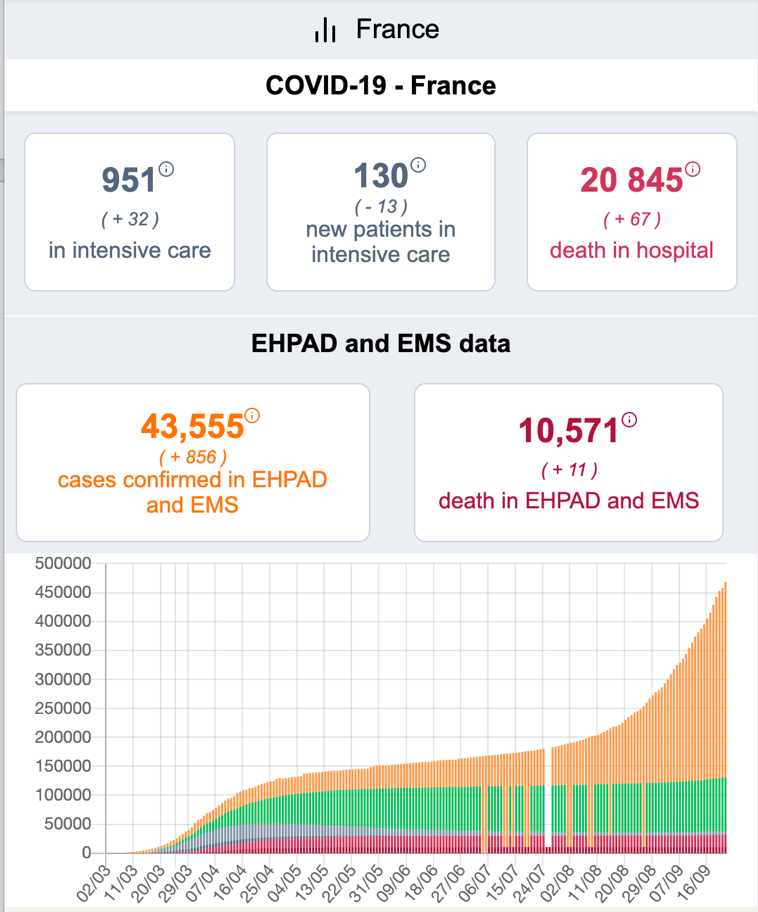 (Chart and data from Public Health France)
(Chart and data from Public Health France)
There have been almost 2.9 million confirmed cases of the virus in Europe and over 186,000 people have died, data from the European Centre for Disease Control and Prevention shows.
What can we learn from the 1918 second spike?
The 1918 Spanish Flu appeared in full force in March of that year, similar to 2020. By summer, the cases slowed. By fall, it appears the flu mutated and by late fall, the so-called second wave hit. History.com recounts:
From September through November of 1918, the death rate from the Spanish flu skyrocketed. In the United States alone, 195,000 Americans died from the Spanish flu in just the month of October. And unlike a normal seasonal flu, which mostly claims victims among the very young and very old, the second wave of the Spanish flu exhibited what’s called a “W curve”— high numbers of deaths among the young and old, but also a huge spike in the middle composed of otherwise healthy 25- to 35-year-olds in the prime of their life.
Look at this CDC graphic of when the most people died in the 1918-19 outbreak. The second wave was worse than the first one, partly because people were indoors during the winter months.
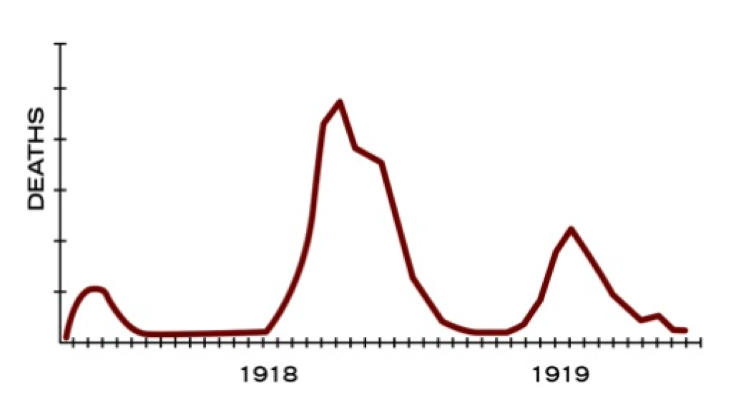 (From CDC.gov: There were three different waves of illness during the pandemic, starting in March 1918 and subsiding by summer of 1919. The pandemic peaked in the United States during the second wave, in the fall of 1918. This highly fatal second wave was responsible for most of the U.S. deaths attributed to the pandemic.)
(From CDC.gov: There were three different waves of illness during the pandemic, starting in March 1918 and subsiding by summer of 1919. The pandemic peaked in the United States during the second wave, in the fall of 1918. This highly fatal second wave was responsible for most of the U.S. deaths attributed to the pandemic.)
Then there was, as you can see, a third wave in 1919. The third wave of the pandemic subsided during the summer of 1919. About 1/3 of the world’s population was infected with the 1918 flu virus – resulting in at least 50 million deaths worldwide.
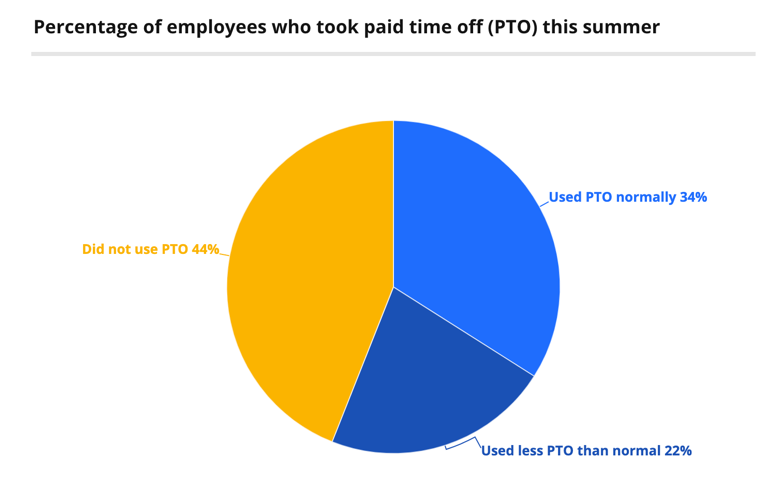
72% of Americans did not take summer vacations during COVID
Yup, this one is me and probably you, too. Forty-four percent of workers have not used paid time off according to Value Penguin’s survey of 1,100 Americans, and your bosses are starting to get worried that you are going to want to take all of your days in the last month or two of the year.
 Younger people were more likely to take their paid time off than their parents or grandparents. In part, people stayed home because they felt they didn’t have any place else to go, but some said they feel guilty taking time off when they are working from home, as if working from home is like being off (it’s not).
Younger people were more likely to take their paid time off than their parents or grandparents. In part, people stayed home because they felt they didn’t have any place else to go, but some said they feel guilty taking time off when they are working from home, as if working from home is like being off (it’s not).
And when job security is far from certain, some of you didn’t want to take time off because you fear it might put you at a greater risk of layoffs and/or furlough. And then there is the matter of not having enough money to take a vacation.
Winter could bring a crisis for people experiencing homelessness
In a typical winter, it is difficult to find enough beds for people who have no home in which to sleep. Now, with the pandemic limiting shelter bed space, the winter ahead could be a stress test.
The Washington Daily Olympian warns that after the better part of a year living under a pandemic, shelters will notice that the people who need a place to sleep and eat will be increasingly tense and maybe more unhealthy.
The Washington State Department of Health predicted when the pandemic began that six to nine months after the COVID outbreak, we would reach the peak of behavioral and mental health issues. We are in that window of time now. KXLY TV (Spokane) reported:
According to the report, “the behavioral health outcomes from COVID-19 for most people are related to experiences of social isolation, fears of the unknowns around further restrictions and economic losses, and stress and pressure related to the balance of childcare and work.”
DOH also warned that as cases climb, there is also a risk of relapses related to addiction.
The way we work now
We’ll be back tomorrow with a new edition of Covering COVID-19. Sign up here to get it delivered right to your inbox.
Al Tompkins is senior faculty at Poynter. He can be reached at atompkins@poynter.org or on Twitter, @atompkins.