Covering COVID-19 is a daily Poynter briefing of story ideas about the coronavirus and other timely topics for journalists, written by senior faculty Al Tompkins. Sign up here to have it delivered to your inbox every weekday morning.
Covering COVID-19 is a daily Poynter briefing of story ideas about the coronavirus and other timely topics for journalists, written by senior faculty Al Tompkins. Sign up here to have it delivered to your inbox every weekday morning.
Once again, one of the leading coronavirus vaccine candidates delivered promising news and the stock market jumped while most everyone celebrated. But there is a lot we do not know about this vaccine.
Moderna’s phase 3 drug trial involves 30,000 volunteers. Half of them took the vaccine and half got a placebo shot. The first report on how the trial is going involves just 95 of those people, so the bulk of the results is still to come. But when results are as strong as Moderna’s it is fairly convincing: The company says its vaccine produced a response that could prevent COVID-19 infections in 90 of the 95 people who got the vaccine.
Moderna’s 94% efficacy rate is in line with Pfizer’s report from a week ago that its own COVID-19 drug has a 90% efficacy rate.
But Moderna’s vaccine has one big advantage over the Pfizer drug: Pfizer’s needs super-cold storage. The Moderna vaccine can be stored for a month at 36 to 46 degrees Fahrenheit, which means drug stores and clinics can store the drug in regular refrigerators.
Here is what we do not know:
- Are these drugs preventing infection or are they just producing antibodies that keep the virus from harming us when we get infected?
- How long will these vaccines last? Will we need a booster shot? How often?
- When will we have a vaccine for children? How diverse was the pool of volunteers? Moderna says its test “included 15 older adults (ages 65+) and 20 participants identifying as being from diverse communities (including 12 Hispanic or Latinx, 4 Black or African Americans, 3 Asian Americans and 1 multiracial).”
- How much of an adverse effect is too much? Moderna says about 10% of the patients felt “fatigue” after taking the vaccine, and a small number experienced some pain and headaches. Those kinds of reactions are not unexpected, but we do not know how much of a negative reaction will be too much and become a reason people might not take the shot.
- What will happen if Johnson & Johnson can produce a one-shot vaccine that is just as effective as the two-dose drugs from Pfizer and Moderna?
- How will people keep track of which vaccine they got if multiple brands become available?
What is next for the vaccine trials?
You might as well learn some new acronyms that you are going to need to know in the weeks ahead as we cover the vaccines’ march through the approval process.
As soon as the drug companies get enough data, they will file an application with the Food and Drug Administration to get emergency use authorization.
VRBPAC: To get the emergency approval, the company presents its findings to the FDA’s Vaccines and Related Biological Products Advisory Committee. “The Committee includes 15 voting members including the Chair,” the FDA said in a report. “Members and the Chair are selected by the Commissioner or designee from among authorities knowledgeable in the fields of immunology, molecular biology, rDNA, virology; bacteriology, epidemiology or biostatistics, vaccine policy, vaccine safety science, federal immunization activities, vaccine development including translational and clinical evaluation programs, allergy, preventive medicine, infectious diseases, pediatrics, microbiology, and biochemistry.”
ACIP: After the VRBPAC reviews the findings, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices weighs in. “The ACIP includes 15 voting members responsible for making vaccine recommendations,” the CDC says. “The Secretary of the U.S. Department of Health and Human Services (DHHS) selects these members following an application and nomination process. Fourteen of the members have expertise in vaccinology, immunology, pediatrics, internal medicine, nursing, family medicine, virology, public health, infectious diseases, and/or preventive medicine; one member is a consumer representative who provides perspectives on the social and community aspects of vaccination.” You can see the members of that committee here. A lot of them are local experts from teaching hospitals and local and state health departments.
VAERS: This is the Vaccine Adverse Event Reporting System. It monitors how vaccines work long after they are administered. VAERS is “a passive reporting system, meaning it relies on individuals to send in reports of their experiences.”
Two more companies, Johnson & Johnson and AstraZeneca, will report their first phase 3 trial results soon. Both companies had to briefly stall their trials to investigate small reports of negative reactions in participants. Those two companies are using a different kind of technology than Pfizer and Moderna, which used a previously untested mRNA method to make their vaccines. AstraZeneca and Johnson & Johnson’s vaccines use genetic material from the coronavirus and an uninfectious adenovirus; technology used to develop an Ebola vaccine.
It was important to not have all of the pharma companies using the same technology in case one worked and another did not.
The probable timeline for vaccines
If (and it is still a big if) the trials keep producing encouraging results, both Pfizer and Moderna could get permission to distribute their drugs in late December. In January, senior citizens, the most at-risk populations (including those in nursing homes, jails and prisons), first responders and health care workers might be inoculated.
The general population would likely be invited to get their shots in the general range of April to June 2021. But that is the most optimistic outlook.
This means it would be late summer at best before we have any real confidence that we might not see a new fall 2021 surge. That would be great news for public schools and universities to try to reopen in-person classes.
Too much optimism?

A health care worker gives a test at a COVID-19 testing site during the coronavirus outbreak in San Francisco, Monday, Nov. 16, 2020. (AP Photo/Jeff Chiu)
Make no mistake, the vaccine news is good news. But there is a bit of danger in hyping the optimism. The pandemic is filling hospitals and threatening school and business shutdowns. This is no time to relax. We have to get through the winter.
It is so telling that work-from-home companies like Zoom, Teledoc, Peloton and GrubHub saw stock prices drop on the Moderna news, while travel companies like Expedia and Carnival saw their stock prices rise.
Stockpiling again

A sign to demote the limit on purchases of paper products hangs on the empty shelve of the paper aisle in a grocery store Wednesday, Nov. 4, 2020, in southeast Denver. (AP Photo/David Zalubowski)
As COVID-19 cases rise, people are getting antsy and stockpiling again. There is no reason to do so, except buying a lot of toilet paper may make you feel like you are doing something.
Seattle supermarkets, for example, are running low on toilet paper supplies.
Supermarket News, an industry news source, reports:
More than half of U.S. consumers are mulling resupplies of groceries and essential items stockpiled at the outbreak of COVID-19, new research from Inmar Intelligence finds.
Winston-Salem, N.C.-based Inmar said Tuesday that 57% of shoppers surveyed are now deciding whether to replenish their stock of goods gathered at the start of the coronavirus pandemic. Meanwhile, another 54% of respondents said they aim to be prepared by always having a stockpile of key items going forward, in case of safety issues and supply outages at stores.
Even though a large percentage of people surveyed said they still have stored-up supplies from the spring, they are adding to it with the predictable stuff:
As in the first COVID-triggered run on grocery stores in March, cleaning and hygiene products are atop consumers’ lists of items to stockpile. The top two products cited by survey respondents were toilet paper (67%) and hand sanitizer (57%), which Inmar said reflects shoppers’ experience of these items often being unavailable early on in the pandemic.
Other products that consumers have stockpiled or plan to stockpile include paper towels (cited by 52% of those polled), disinfecting wipes (53%) and canned goods (54%). And 45% of respondents said the second round of stockpiling will include items not in the first round, such as frozen dinners, pasta, snacks and cleaning products.
Some stores are starting to set limits on how much you can buy of certain items.
And it may just be that we are storing better quality stuff than last time. CNBC reports:
In recent weeks, however, consumers have gravitated toward premium purchases such as higher-end coffees, cheeses and frozen entrees instead of value brands or private labels, market research firm IRI said.
Sales of premium and superpremium packaged goods grew by 1.7% at retailers year over year for the 26 weeks ended Oct. 4, according to IRI data. The percentage may sound like a small one, but it is a notable shift in the multibillion-dollar consumer packaged goods industry, where rivals compete at just tenths of a percentage from one another.
A surge in the spice industry
This might make 2020 seem like a throwback to, say, the 1400s, when spices were the precious hub of international trade. But spice companies say since we are cooking at home more these days, we are using more spices and herbs and the demand is great enough to make up for all of the restaurant sales that slumped. The Washington Post notes:
Sales of McCormick & Co.-branded spices and herbs were up 35 percent over last year for the 13 weeks ending Aug. 30, according to Information Resources, a data analytics firm. One of the world’s largest spice companies, based in Hunt Valley, Md., is up nearly 16 percent in dry recipe mixes in that same time frame, nearly 10 percent for mustard, 44 percent for hot wing sauce and 35 percent for barbecue sauces, the data shows.
Buyers’ willingness to pay a premium for new flavors and global tastes has been fueling the growth of the international product market for some time. According to Grand View Research, the global seasoning and spices market was valued around $13.8 billion in 2019 and was expected to see substantial growth through 2027.
The pandemic has accelerated this. According to the NPD Group, national consumption of spices, seasonings, marinades and rubs was up over 50 percent in July 2020, the most recent month for which data is available, compared with July 2019. Much of the increase has been seen at breakfast and lunch, two meals historically eaten away from home. Instead of the grab-and-go staples of the pre-pandemic era, households have more time, and breakfasts have become more involved. Home lunchers have upped their condiment and seasoning game.
A nagging glass shortage
The spice companies have complained, however, about a shortage of glass containers and lids. They are not alone.
From solar panel manufacturers to beverage companies, a glass shortage that has been running the length of the pandemic is still clogging supply chains. Some construction projects reported glass prices have spiked in the last year.
What we learn from an island of low COVID-19 rates in a state of infections
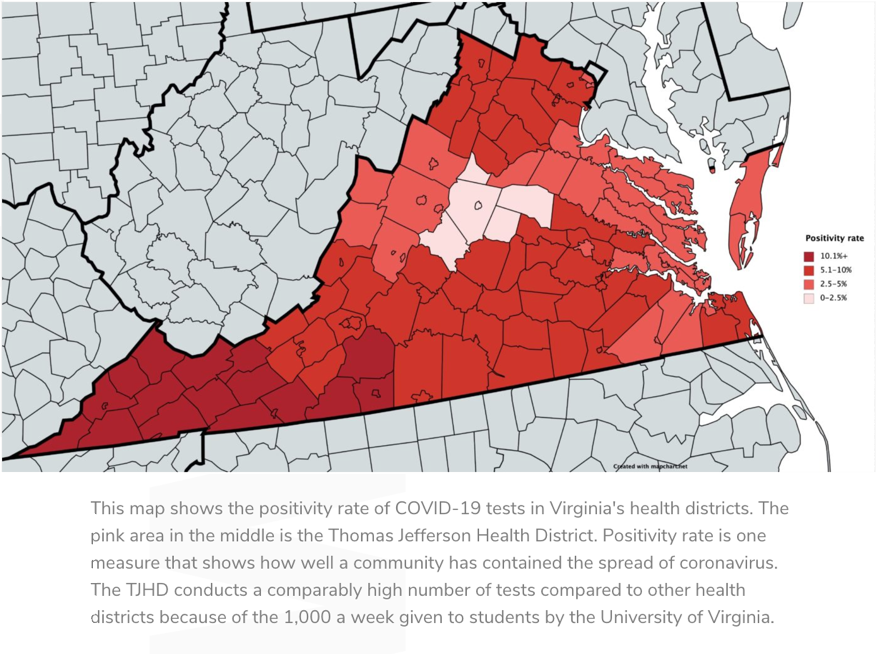
(Charlottesville Tomorrow)
Charlottesville Tomorrow puts the situation this way: “While coronavirus cases in the past weeks have spiked across Virginia and elsewhere in the country, the trend in the Charlottesville-area has been quietly moving in the opposite direction.”
The positive test rate around Charlottesville has hovered around 2% while the rest of the state is three times that. Health experts cannot point to any one thing that accounts for the difference, except people in Charlottesville appear to be more willing to wear masks and The University of Virginia, the local university, has been vigilant since cases began to rise this fall.
I point to this story as a way to show that posting maps about how bad the virus has spread is not the only way to cover the story. Find examples of successes, even modest successes, and find out what those communities are doing differently.
Of course, it will only take one Thanksgiving gathering or one frat party to upend a hopeful trend, so don’t sugarcoat the situation.
The Charlottesville community is in what is called the “Thomas Jefferson Health District,” and this is the trend map every community would wish for:
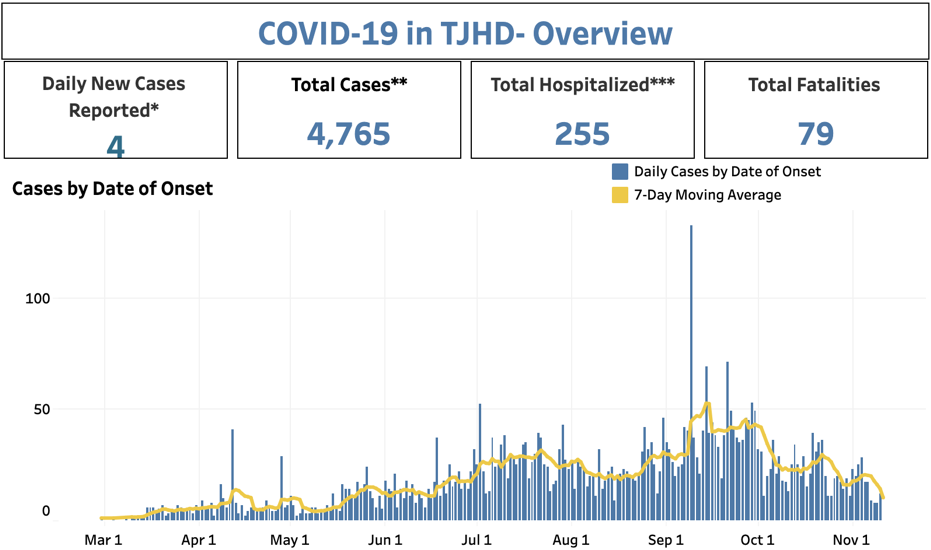
From the TJHD data portal, which is updated daily (Virginia Department of Health)
Charlottesville health providers say they are still trying to find ways to address the fact that despite the overall lower COVID-19 rate, Black patients make up 13.7% of COVID-19 cases there.
“The difference is even more stark in the Latino community: making up 16.5% of all cases despite being just 5% of the district population,” WVIR-TV says. “The rate of severity is also much higher among Black patients: They make up 34.3% of hospitalizations and 22% of deaths.”
Those are the kinds of stats that give journalists a target on which to focus some intensive coverage.
Many stores will not open on Thanksgiving this year

Shoppers walk past a display of Christmas decorations at a Target store, Sunday, Nov. 8, 2020, in Marlborough, Mass. (AP Photo/Bill Sikes)
For the first time in years, in-person Black Friday shopping, such as it is in 2020, will begin on Black Friday. Lots of stores — including Walmart, Best Buy, Lowe’s, Sam’s Club — say they will be closed on Thanksgiving. You don’t have to wait. There are lots of early deals on now.
Longer Zoom meetings are not necessarily good news
Zoom is ruining the best excuse you have to bail out on a call after 40 minutes. On Thanksgiving Day, Zoom is lifting the 40-minute limit on free calls, so your calls can go on and on and on. Zoom probably knows it will take much of the 40 minutes to get people to unmute and not have the laptop camera aim up their noses.
The way we live now
Here comes Santa Claus – with face masks and plexiglass: https://t.co/b3Lxua7KIb pic.twitter.com/2AHqEdtAsS
— NBC DFW (@NBCDFW) November 16, 2020
We’ll be back tomorrow with a new edition of Covering COVID-19. Sign up here to get it delivered right to your inbox.