Covering COVID-19 is a daily Poynter briefing of story ideas about the coronavirus and other timely topics for journalists, written by senior faculty Al Tompkins. Sign up here to have it delivered to your inbox every weekday morning.
Covering COVID-19 is a daily Poynter briefing of story ideas about the coronavirus and other timely topics for journalists, written by senior faculty Al Tompkins. Sign up here to have it delivered to your inbox every weekday morning.
No doubt you have seen the advertisement that President Donald Trump is running claiming he has lowered drug prices. Back in 2016, he promised to lower drug prices and, in this reelection campaign, he is claiming success.
On July 24, President Trump held a signing ceremony that was portrayed as imposing a “favored nations clause” on drugs sold in the United States. That means that the price of some drugs would be tied to the low prices that some foreign countries pay.
The president signed four orders on that July day and called the “favored nations” order “the granddaddy of them all.”
But there is a catch. As far as we can tell, there is no such order in place. The New York Times explained:
The president also said he would meet with executives the week after the signing ceremony. Such a meeting never occurred. However, one person familiar with the issue said the White House had been meeting with the pharmaceutical manufacturers’ trade association to try to come to terms on a joint statement.
In the meantime, Mr. Trump has been boasting about his plans as if they were in place and drastically affecting drug prices. His campaign has spent $668,000 over 11 days on a new advertisement suggesting that his Democratic opponent, former Vice President Joseph R. Biden Jr., is the favored candidate of the drug industry, attacking the companies and Mr. Biden in one fell swoop.
When he signed the document, the president said he would give drug companies a month to lower prices. Trump said, “We’re going to hold that until Aug. 24, hoping that the pharmaceutical companies will come up with something that will substantially reduce drug prices.”
And he promised that no matter what the drug companies do, the prices are coming down, “And the clock starts right now. So, it’s Aug. 24 at 12 (a.m.), after which the order on favored nations will go into effect.”
He has kept that message up, tweeting:
A “Favored Nations Clause” against Big Pharma, which I signed last week, means that the USA gets a price on Prescription Drugs that matches the price of the Country that pays the lowest price anywhere in the World. 50%, 60%, maybe 70% reduction. No other President would do this!
— Donald J. Trump (@realDonaldTrump) August 23, 2020
That Aug. 24 deadline has passed and the president claims the order as a victory, but as The Hill put it:
That deadline has now come and gone, without the announcement of any deal with drug companies, yet the White House has not moved forward with the order and is not saying if it will.
You can see, by going to the White House website, the administration posted three of the four orders signed on July 24. But not the “favored nations” order.
What exactly did the president sign? What did the order say? We don’t know, and the White House won’t say.
As he signed those documents in July, the president held the document up for cameras. That is as close as we have come to seeing what is in the order. Modern Healthcare tried to make out what the document says. The part that you can see in the photograph makes a mention of the need for rule-making, which is way different from ordering a change in drug pricing, which the president cannot do alone anyway.
More than that, if it actually was a signed executive order then, by law, it should be public. The Times story said:
Phillip J. Cooper, a professor of public administration at Portland State University and the author of “By Order of the President,” a book about presidential documents, said that federal law required signed executive orders to be published in the Federal Register. No “favored nation” order has appeared, so the missing order either violates the law or is incomplete.
The story for journalists is to keep asking for the documents and to point out that the claims about drug prices are, at minimum, premature.
One voice you can turn to is Patients for Affordable Drugs Now, which is pressing the president to move forward with his promise.
The president’s plan included a “benchmark” for Medicare costs against the pricing of drugs in 16 other nations as part of an international pricing index.
The Pharmaceutical Research and Manufacturers of America said a “favored nations clause” would, in effect, give other countries the power to set drug prices for Americans. The drug manufacturers favor “market-based, competitive solutions.”
FDA commissioner says he misspoke about plasma effectiveness
In D.C. parlance, Food and Drug Administration commissioner Stephen Hahn “walked back” his enthusiastic endorsement of COVID-19 plasma treatments to say that if current plasma tests continue to show what limited tests have shown then it might be useful in treating the coronavirus.
That is a long way from Sunday’s declaration that it is a “breakthrough” treatment. Hahn announced an emergency approval for doctors and hospitals to use COVID-19 plasma in a Sunday evening news conference with the president.
He made the correction on Twitter, saying, “I have been criticized for remarks I made Sunday night about the benefits of convalescent plasma. The criticism is entirely justified. What I should have said better is that the data show a relative risk reduction not an absolute risk reduction.”
The Mayo Clinic, which oversees the plasma treatment tests, is using blood plasma from people who have tested positive for COVID-19 to infuse into critically ill COVID-19 patients. The first round of data showed that when critically ill patients were given plasma within three days of diagnosis of COVID-19, 8.7% of them died. But if the treatment was delayed until four or more days, the fatality rate was 11.9%. At 30 days, the mortality rate was 22% for the early-treatment group versus a 27% mortality rate for the people who got later treatment.
NPR said for some in the medical research world, the Twitter correction is not enough:
Some of Hahn’s critics are not satisfied by his apology. “Serious mistakes undermine your credibility,” tweeted Dr. Eric Topol, a high-profile researcher at the Scripps Research Translational Institute. “Sorry your (twitter thread) doesn’t cut it,” Topol continued. “When will you have a press conference to fully correct this? And fix FDA’s website? To restore public trust. Please.”
Jeremy Berg, the former editor of Science magazine, chastised Hahn as well. “Be more careful with your language next time,” he wrote on Twitter. “This really matters for the public’s understanding of COVID-19 and of science.”
The pity of this confusion is twofold. It undermines trust in the FDA just when we need to be able to trust it the most. It may also undermine what still could be a useful tool in treating the virus. Blood banks are enthusiastically collecting plasma from donors who have recovered from COVID-19 in order to keep the study going.
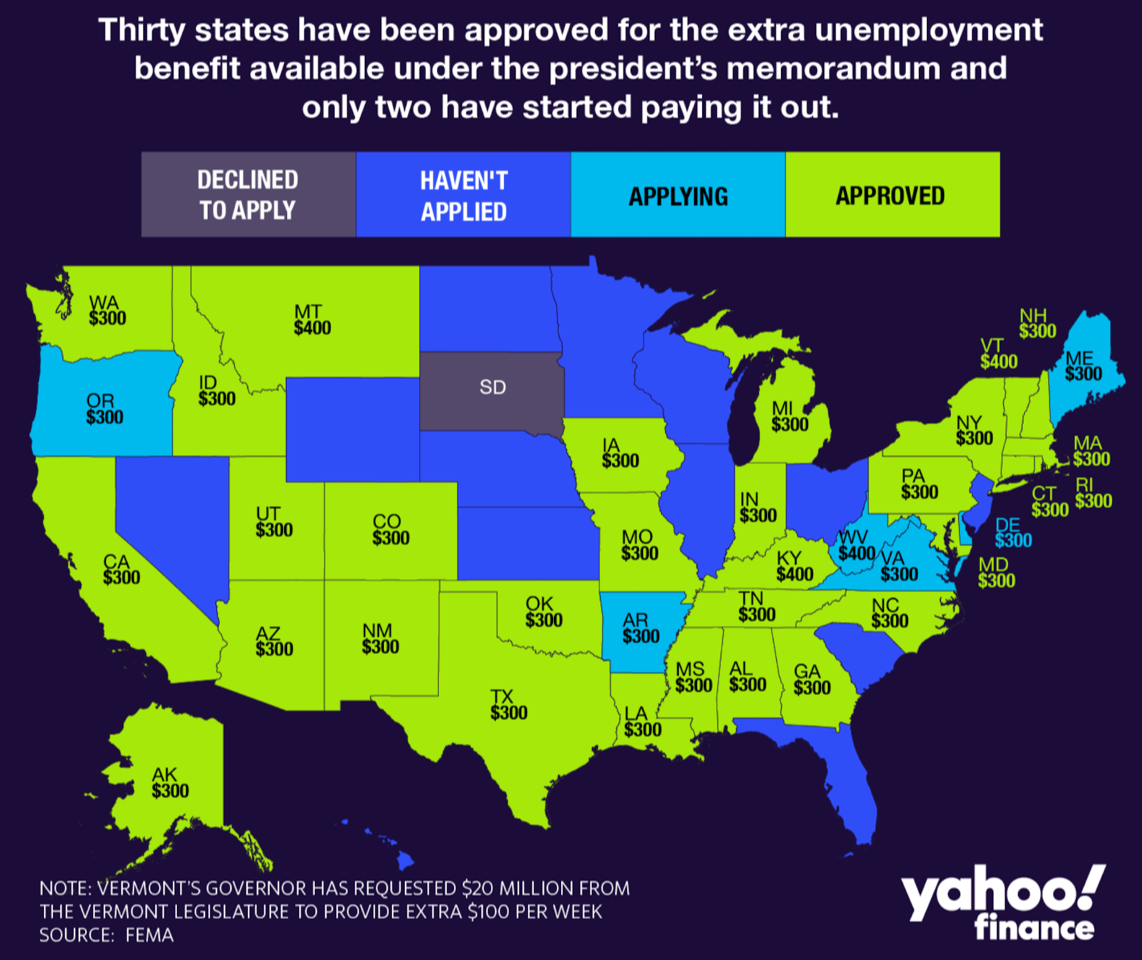
Some states have started paying the $300 federal unemployment. Most haven’t.
Seven states are not waiting for Congress to come to an agreement on a new federal stimulus bill. They are grabbing President Trump’s offer for a $300 federal unemployment payment paid out of the Federal Emergency Management Agency’s budget.
Arizona, Iowa, Louisiana, New Mexico, Colorado, Missouri and Utah were the first states to ask for and receive approval for FEMA funds to send out as unemployment payments. The payments were initially described as a $400 payment — of which states would pay $100 — but when states balked, the feds said a $300 payment could go on with no match.
By Tuesday evening, the list of states that are clear to distribute the $300 payments quickly grew to 30. (See announcements for each state here.) Three states — Kentucky, Montana and West Virginia — are opting to pay the full $400.
States approved for aid are guaranteed just three weeks of funding, with payments backdated to the week ended Aug. 1. However, they may get more aid, depending on how many states apply and how quickly the money is drawn down.
Not all unemployed workers are eligible for the payments. Those currently getting less than $100 a week in unemployment benefits won’t receive the assistance — amounting to thousands of people, and perhaps more than 1 million.
Rent payments keep dropping and evictions loom
Those unemployment checks cannot arrive soon enough. The latest data about how renters are paying their bills shows tough times ahead. Rentec Direct provided this data:
As of Aug. 10, rent payments received nationwide by property managers and landlords are 29% lower than rent received for the same period in March, prior to the onset of the COVID-19 pandemic in the U.S.
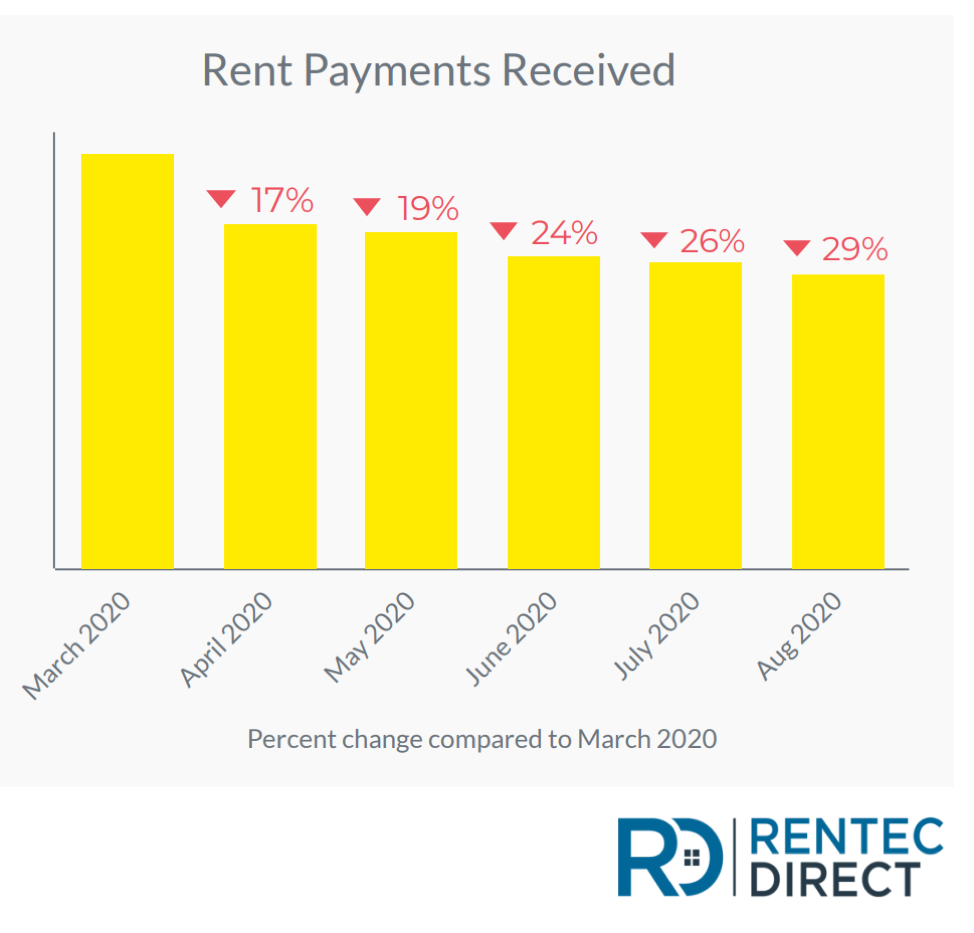
(From Rentec Direct)
Reuters took a look at New Orleans, where this week marks 15 years since Hurricane Katrina devastated the city.
New Orleans was battered here early by the coronavirus, and as tourism shut, nearly one in five residents were put out of work in April, according to the Bureau of Labor Statistics.
As the city slowly tries to reopen, that dropped to 12.9% in June, but many people are still trying to catch up to lost coronavirus income, advocates say. Up to 56% of Louisiana’s renters are now at risk of eviction, the Aspen Institute calculates here, the second-highest percentage of at-risk renters in the country after Mississippi.
The New York Times reported this week:
In Nebraska, where the local moratorium on evictions expired in May, at least 92,000 people are at risk of being forced out of their homes in the coming months, according to a report by a consortium of housing advocates and public policy organizations. The report said that nationally, at least 30 million people — including those in homes not covered by the CARES Act moratorium — were in danger of being evicted without any new federal aid or a renewed pause.
Two emergencies will strain relief agencies
The American Red Cross said it is looking for people to help with shelter relief efforts as it is responding to California wildfires and a growing hurricane heading for the Louisiana-Texas coast.
Volunteers from Maine and Oregon are heading to California, while Florida and Texas volunteers are heading for the hurricane’s path.
Because of the pandemic, volunteers don’t check in for duty. Instead, the Red Cross dispatches them virtually.
Usually, after a hurricane, good-hearted people with boxes of supplies, hammers and plywood stream in to lend a hand. But in a pandemic, everything is more complicated.
FEMA offered advice on how to help:
Donate Money and In-Kind Goods
- Cash donations to the non-profit of your choice is the best way to help.
- You can find vetted non-profit organizations supporting COVID-19 response efforts at NVOAD.org.
- Don’t collect or make donations until you are sure it is needed, who will accept it and how it will get there.
- Email FEMA-Donations-MGT@fema.dhs.gov with questions about how best to donate.
Donate Blood or Plasma
- Another vital need during this time is blood and plasma donations. Unfortunately, many blood drives have been cancelled and donations at plasma collecting centers have significantly declined.
- Donating blood and source plasma is a safe process. Both blood collecting centers and plasma collecting centers have the highest standards of safety and infection control
- To find where you can donate blood, visit aabb.org. To find where you can donate plasma, visit the FDA’s Donate COVID-19 Plasma page.
Volunteering: Licensed Healthcare Professionals
Find information on eligibility, view credential levels by clinical competency and register with the Emergency System for Advance Registration of Volunteer Health Professionals in their state.
Volunteering: Medical Reserve Corps Volunteers
Contact an MRC unit in your area to help your communities with call centers, drive through clinics and more.
Donate Supplies and Equipment
If you represent a business that would like to donate medical supplies, equipment or pharmaceuticals, please provide details on what you would like to give. This form is for donations only.
We’ll be back tomorrow with a new edition of Covering COVID-19. Sign up here to get it delivered right to your inbox.
Al Tompkins is senior faculty at Poynter. He can be reached at atompkins@poynter.org or on Twitter, @atompkins.