Covering COVID-19 is a daily Poynter briefing of story ideas about the coronavirus and other timely topics for journalists, written by senior faculty Al Tompkins. Sign up here to have it delivered to your inbox every weekday morning.
Covering COVID-19 is a daily Poynter briefing of story ideas about the coronavirus and other timely topics for journalists, written by senior faculty Al Tompkins. Sign up here to have it delivered to your inbox every weekday morning.
The Centers for Disease Control and Prevention is modeling how the weeks ahead will look. They look pretty grim. I want to share some charts that were produced by researchers at the University of Texas at Austin. Look at them and then I will supply some details on what they tell us.
Here are the scenarios that the graphics spell out. Notice all of the models show a peak around late January or early February. The first charts say a lot of people are going to get sick. The second charts say tens of thousands will be hospitalized. The third charts predict that the peak of deaths will happen in February, a couple of weeks after we hit peak infections. The worst case might involve between 4,000 to 5,000 deaths a day. The best case would be about half that much.
Epidemiologist Dr. Katelyn Jetelina explains:
Best case scenario (purple line below; scenario B): By mid-January 190,000 people catch the virus every day—about double what the case rate is today. In this scenario, Omicron would lead to 10,500 hospitalizations per day (a few thousand more than today) and 1,400 deaths (a few hundred more than today).
Worst case scenario (pink line; scenario C): By January more than 500,000 people would catch the virus every day, which is more than double the peak reached last winter. 30,000 people would be hospitalized per day and 3,900 would die every day. This scenario is the most pessimistic and, in my opinion, won’t happen for two reasons:
This model assumes that Omicron is more severe than Delta. This is not the case; in fact, there is considerable debate as to whether Omicron is less severe.
This model also assumes that there is no behavior change, which is also not realistic. People change their behaviors when cases increase, whether they realize it or not.
I compared those projections with the newest estimates from the Institute for Health Metrics and Evaluation at the University of Washington. The trend lines are very similar:
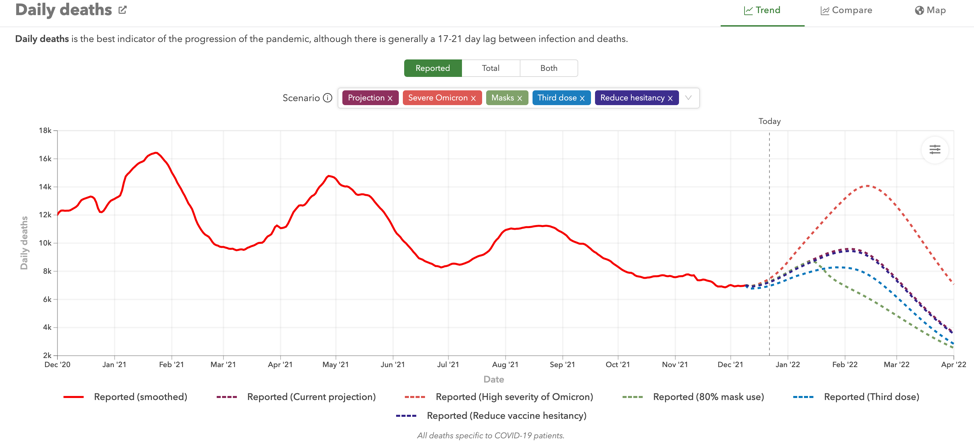
(IMHE)
The newest IMHE projections show hospitalizations peaking in mid-February at about the same rate as we saw last summer during the peak of the delta wave:
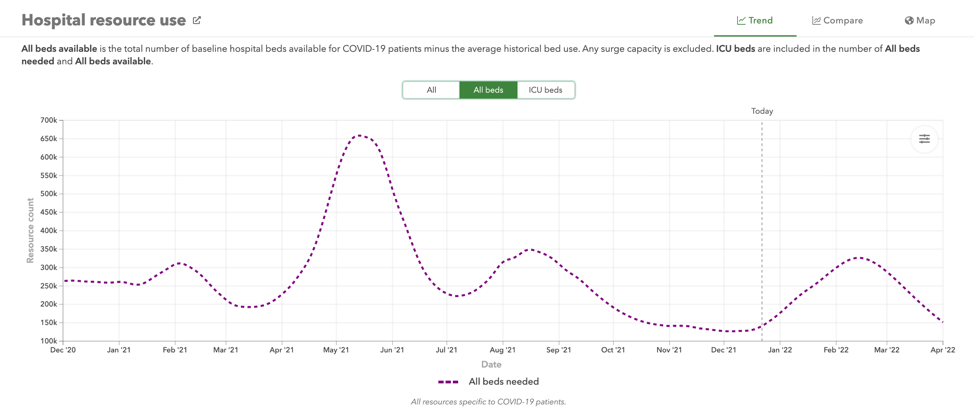
(IMHE)
Outdoor concert becomes an omicron super-spreader

Bad Bunny performs “Lo Siento BB :/” at the American Music Awards on Sunday, Nov. 21, 2021, at Microsoft Theater in Los Angeles. (AP Photo/Chris Pizzello)
Until now, Puerto Rico has led the rest of the United States in vaccination rates and low COVID-19 infection rates. But an outdoor Bad Bunny concert there may be connected to at least 2,000 cases.
I emphasize, it was an outdoor concert. Those in attendance had to show proof of vaccination. Some are pointing out that there have been a lot of holiday gatherings in the last week or so and the island’s positive test rate has been rising. But, wow, look at this chart:
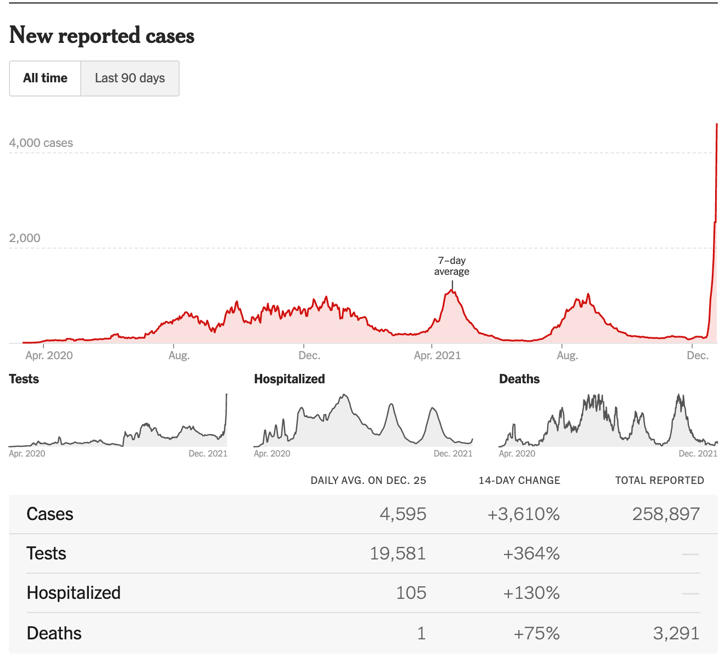
(The New York Times)
Journalists, when we look at the figures from Puerto Rico, there is a lesson in context for you to consider. First, do not become entranced by percentages. The positive test rate does not tell us the severity of the illnesses. And it would be easy to take Puerto Rico as an example of the idea that vaccines are not preventing COVID-19. But look beyond the positive test figures at hospitalizations and deaths. While they are up as a percentage — because the previous figures were quite low — the daily number of deaths is 1. Even that is a 75% increase. It is likely that we will find that Puerto Rico’s high vaccination rate will keep the island’s death rate from jumping and that will be a testimony to the vaccines’ effectiveness.
Who is making money from all of this testing?

People leave a free COVID-19 PCR and Rapid testing site in Glenview, Ill., Saturday, Dec. 25, 2021. (AP Photo/Nam Y. Huh)
The Kaiser Health News podcast “An Arm and a Leg” investigated why at-home COVID-19 test kits are so expensive. Kaiser talked with Eric Umansky, an editor at ProPublica, and his colleague Lydia DePillis, who found that other countries did two main things the U.S. did not.
First, other governments bought boatloads more test kits than the American government did. Second, other governments approved more tests, so there is some market competition. As a result, test kits in Europe are more than half as expensive as the same tests in the U.S.
ProPublica offers additional insight into why the United States has expensive tests that are hard to find:
FDA officials said they simply want to ensure that rapid tests detect even low levels of the virus, since false negative test results could cause people to unwittingly spread the disease. They blame the test shortages on an absence of the kind of sustained public funding that European governments have provided. Without it, manufacturers have lacked confidence that going through the FDA’s process would be financially worth the trouble.
“Where we have seen tests truly coming to the marketplace, the big difference has been government investment,” said Dr. Jeff Shuren, head of the FDA’s Center for Devices and Radiological Health, which authorizes tests. “Folks will come and do larger volumes because you’re supporting production, which can also help drive down prices.”
Both the Trump and Biden administrations banked on vaccines putting a swift end to the pandemic, holding off on large-scale purchases of COVID-19 tests that Americans could keep in their medicine cabinets.
As a result, one of the few companies that has successfully gotten tests authorized and onto shelves — Abbott Laboratories — has dominated the market. Its BinaxNOW tests account for around 75% of U.S. retail sales, according to data from NielsenIQ, even though they’re sold here for several times the price of the same Abbott tests in Europe.
ProPublica also has a story about an MIT researcher who — very soon after the pandemic began — produced an at-home COVID-19 test that would have cost less than $10. But the U.S. Food and Drug Administration took longer than many thought it should have to approve tests. The FDA says the approval process was slow because developers didn’t provide enough data or, at times, inaccurate data.
Whatever the truth is, you could have had easy-to-find inexpensive home tests, as other countries have.
FDA approved a COVID-19 pill that has a modest track record and some other problems
Everyone wants a pill that can keep people from getting seriously ill from COVID-19. That’s a given. But the FDA just approved a monoclonal antibody for emergency use that has a modest track record of success and a list of issues that might give you pause.
The drug trials for this new pill so disappointed health officials in France that the government canceled its order for a batch of it. Israel is going ahead with deploying it. Read the drug trial info here. The FDA does not recommend the pill for pregnant women or anyone under age 18.
Read this passage from The Washington Post, and pay attention to the stunning first sentence.
Molnupiravir might create new variants of SARS-CoV-2 that evade immunity and prolong the pandemic.
The problem with molnupiravir lies in its mechanism of action. Unlike any previous antiviral drug, molnupiravir does only one thing: It introduces mutations into the viral genome. We are already familiar with the fact that viruses naturally mutate to evade immunity; the many mutations of the spike protein in omicron, for example, allow it to evade the antibodies created by prior infections or vaccines.
Molnupiravir relies on inducing even more mutations so that eventually the virus’s proteins are damaged beyond function. That molnupiravir can mutate SARS-CoV-2 to death has been demonstrated in the controlled conditions of a petri dish and lab animal cages, leading Merck to test it in covid-19 patients in clinical trials.
But people are not petri dishes or lab animals, and while molnupiravir works to some extent, it has not worked very well in covid-19 patients. Specifically, molnupiravir reduced hospitalizations by only 30 percent. In contrast, Pfizer’s antiviral drug Paxlovid, which works by a different mechanism and was also approved by the FDA, reduced hospitalization by 89 percent.
A critical blood shortage
The Red Cross, America’s Blood Centers and the Association for the Advancement of Blood and Biotherapies released a statement that says:
If the nation’s blood supply does not stabilize soon, lifesaving blood may not be available for some patients when it is needed.
This is a warning at the top of the Red Cross website:
There are lots of reasons that blood supplies are thin right now. People are not donating at the rate they did before the pandemic, and blood centers are short on staff. All of the usual stuff. But this is serious.
The Los Angeles Times reports:
The U.S. is currently in the middle of what has been referred to by some as a blood emergency — the country’s blood supply is at its lowest level in a decade. According to Dr. Ross Herron, division chief medical officer at the American Red Cross, the U.S. has been experiencing low donor turnout ever since the COVID-19 Delta variant began spreading last summer, and the inventory of some blood types has dropped below one day’s supply in recent weeks — that’s a dangerously low level.
“We tend to assume that blood is always available in the U.S., however, the only source is volunteer blood donation. COVID has placed significant additional barriers to obtaining blood for emergencies, and for many children and adults who depend on blood transfusion to survive,” explained Dr. Thomas Coates, head of hematology at the Cancer and Blood Disease Institute at Children’s Hospital Los Angeles.
The Red Cross has had less than a day’s supply of certain blood types in recent weeks. The supply of types O positive and O negative blood, the most needed blood types by hospitals, dropped to less than a half-day supply at times over the last month — well below the ideal five-day supply. There is also an emergency need for platelets, which is the clotting portion of blood and must be transfused within five days of donation.
Air Force denies 2,130 religious exemptions for mandatory vaccination
Each branch of the military is supposed to “accommodate a sincerely held belief without putting mission accomplishment at risk,” but applicants for exemptions are finding it difficult to get approval.
The Air Force has turned down more than 2,000 religious exemption requests. 8,600 more requests are pending. Zero have been approved so far.
The Navy said it did not approve any of the 2,844 requests for religious accommodation.
After the pandemic ends, the DOJ may still allow federal prisoners to serve time at home
Around 8,000 people who were released from federal prisons during the pandemic will not have to go back to prison when the pandemic officially ends, the Department of Justice says. In all, somewhere around 36,000 people who were serving time in prisons were released to home confinement. Some have already returned to prison for rules violations. Others served out their sentences. But 8,000 would have had to return under the provisions of their release during the Trump administration. Attorney General Merrick B. Garland said in an official DOJ statement;
Thousands of people on home confinement have reconnected with their families, have found gainful employment, and have followed the rules. In light of today’s Office of Legal Counsel opinion, I have directed that the Department engage in a rulemaking process to ensure that the Department lives up to the letter and the spirit of the CARES Act. We will exercise our authority so that those who have made rehabilitative progress and complied with the conditions of home confinement, and who in the interests of justice should be given an opportunity to continue transitioning back to society, are not unnecessarily returned to prison.
Journalists, in 2022, recognize that we do not have answers for all of the COVID questions
Over the weekend, an opinion piece from Washington Post columnist David Von Drehle really struck me. Here is a passage I want you to see:
The promise of 24/7 news and analysis has heightened and reinforced a natural desire to have perfect knowledge, even in imperfect situations. There’s an expectation that everything one needs to know can be known — right now. Not just the what, but the why and the who and most of all — the what next. This sense of urgency encourages speculation. It nudges us to generalize from isolated anecdotes.
We can safely say, at this point in the pandemic, that there has never been a day in which answers concerning covid-19 outnumbered the remaining questions.
It would have been useful, I believe, to have emphasized the uncertainty — as the best reporters always did. The most knowledgeable voices during this pandemic have been scrupulous about the limits of their knowledge. The public might have been less surprised, less chagrined, less exhausted by changes in public health guidance and contradictory scientific findings. Possibly, there would be fewer people today willing to believe that normal fits and starts of the learning process were somehow signs of bad faith.
Yet this era also discourages humility about the limits of our knowledge while encouraging worship of Big Data. These pandemic years have been choked with data, much of it useless.
It is a useful read. Spend some time with it and let’s all try to keep his words in mind in 2022.
We’ll be back next Monday with a new edition of Covering COVID-19. Are you subscribed? Sign up here to get it delivered right to your inbox.